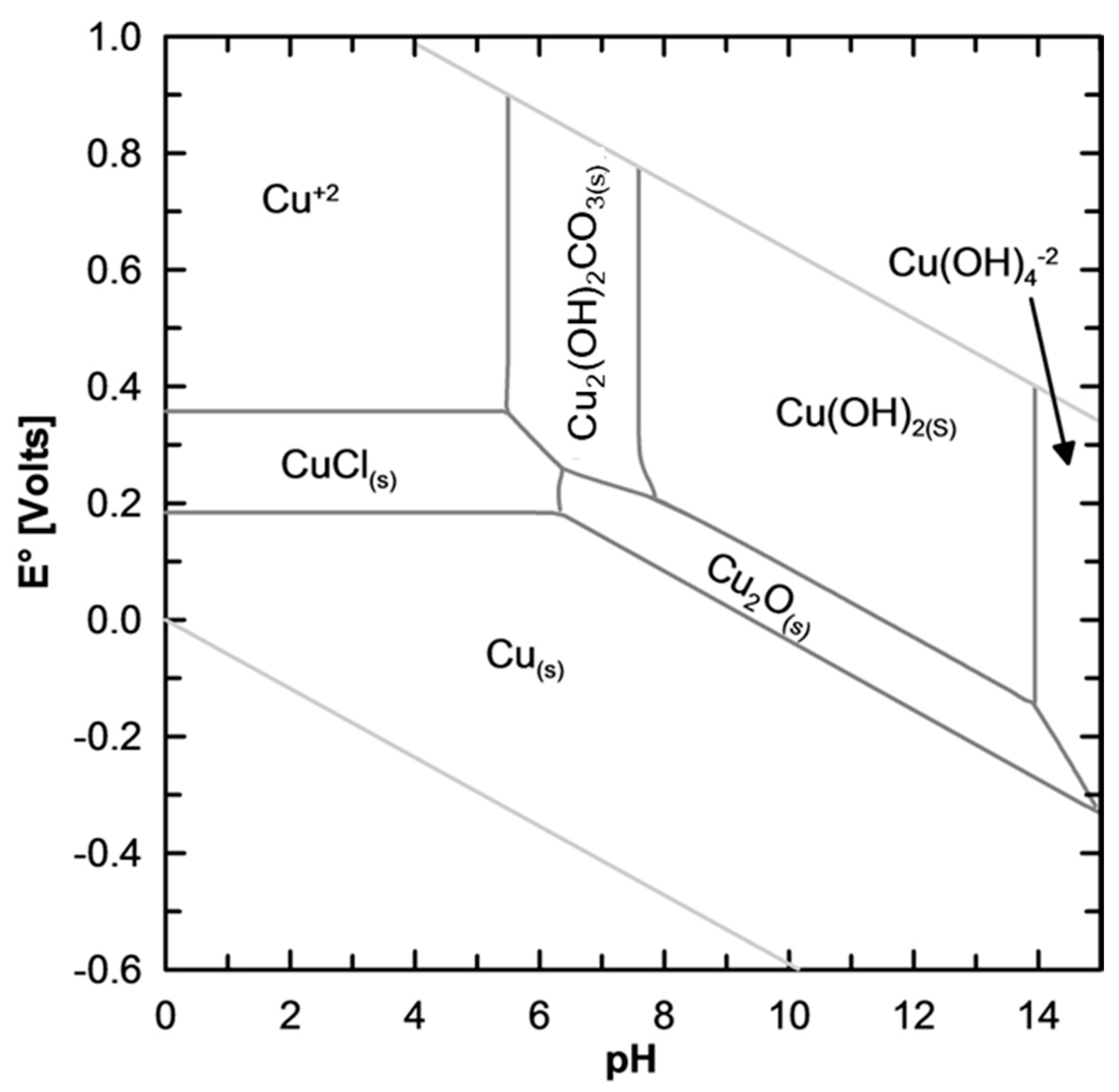
?.6 Draw the two lines corresponding to the redox couples of water on the Pourbaix diagram of copper (E°(O./HzO) 1.23 V). Calculate the corresponding solubility product. II.5 The vertical segment between domains 2 and 3 corresponds to a pH of 7.2. II.4 Determine the slope of the border between domains 1 and 2. A.s Pourbaix diagram fos r copper at temperatures below 25 ☌ are lacking in the literature a, low temperature (5 ☌) diagram has also been produced in this study. II.3 Identify the species corresponding to each of the domains 1-8. The species represented in this diagram are (using simplified formula which correspond to the removal of water molecules) HCuo, Cuo, CuzO, Cu', Cuo, Cu, Cu,O, Cu IL. 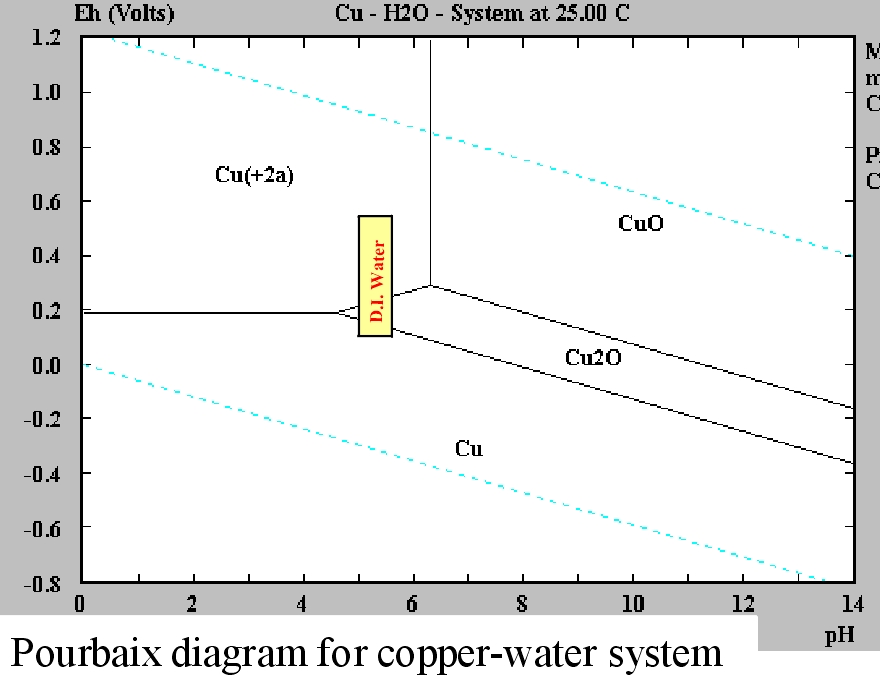
The extraction of three metals (cobalt, copper, and gold) is used to demonstrate the use of these variables, and the impact of assumptions made during their construction. Give the complete formula and name of the eight species included in the diagram. Eh-pH diagrams (aka Pourbaix diagrams, electrochemical phase diagrams) are important to the development of hydrometallurgical and electrometallurgical processes. The species represented in this diagram are (using simplified formula which correspond to the removal of water molecules) HCuOi, Cuo2, Cu-Os, Cu, CuO, Cu', Cu0, Cu IL.1 Give the oxidation number of copper in each of the species included in the diagram II.2 All soluble species in the diagram have an octahedrally coordinated copper ion bearing water or hydroxide ligands only. Transcribed image text: Problem II - Pourbaix diagram of copper The Pourbaix diagram of copper for concentrations in soluble species equal to c10molL is given next page.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
